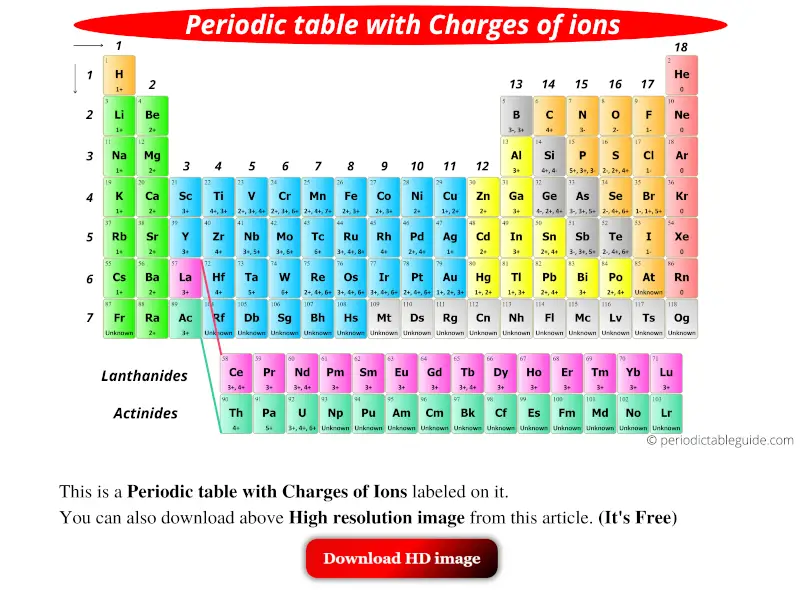
Please re-write this article to improve it to explain concepts before they are used in answers or taken for granted as prior knowledge when they have not first been defined in Unit 1. The problems are then for ionic compounds that are not monoatomic. The use of magnitude is used without being introduced followed by a statement that isn't well described, 'The magnitude of the charge is equal to the number of electrons lost, which is equal to the number of valence electrons in the neutral atom.'Īluminum is in group 13, or IIIA is stated when no introduction has been provided to what IIIA means. 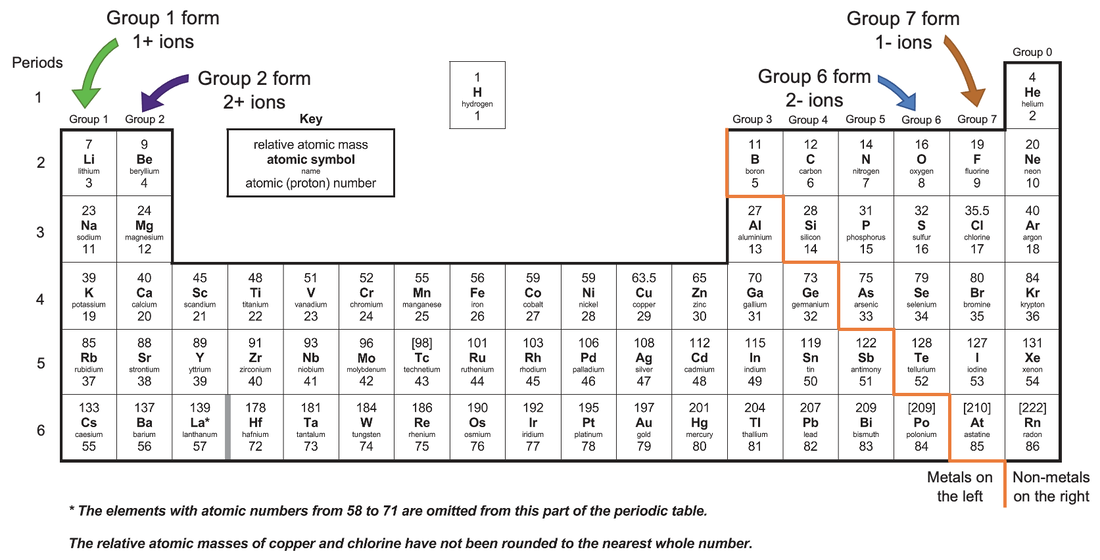
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton. An ion ( / a.n, - n /) 1 is an atom or molecule with a net electrical charge. Forming an ionic bond, Li and F become Li + and F ions. On the Periodic Table metals (found on the left of the table) will be. Electron transfer between lithium (Li) and fluorine (F). They are sufficiently stable that they don’t form either anions or cations easily. Where in Unit 1 is s, p, d, f and shell and subshell defined prior to this article. To find the ionic charge of an element youll need to consult your Periodic Table. To put it another way, elements on the left side of the periodic table tend to form cations, while those on the right side form anions. IUPAC group number is also not described, it is simply used, without definition or prior description.Įlectron configuration is used as part of a hidden explanation as an answer, yet the article or Unit 1 does not explain what an electron configuration is. Where in Unit 1 is Octet and Valence given definition. Use a periodic table to determine the charges achieved upon ionization of main group elements. The common ionic charges of the elements are shown in the table below. Octet is used without definition, followed by valence also being used without definition. State the charge pattern for main group element ionization. List of elements with their common ionic charges. Thus, the periodic table becomes a tool for remembering the charges on many ions. 
We are being asked to 'Remember that periodic groups refer to columns on the periodic table, whereas rows are known as periods' when this concept has also not been initially defined in this article or in Unit 1. In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge. Where is monatomic defined in this article or in Unit 1. An iron ion with a charge of +8 is not very likely, therefore, the octet rule is not applicable to transition elements.Words are used without first having had a definition or explanation in the article or earlier in Unit 1. shows the charge of the metal ions in that compound. In order for an element such as iron (Fe) to achieve the same noble gas configuration of argon (Ar), it would need to lose 6 electrons in the 3 d subshell and 2 electrons in the 4s subshell. The number in the name of the compound close compound A substance formed by the chemical union of two or more elements. As discussed in class, the elements in the first column of the periodic table (excluding hydrogen) always lose one electron to get a +1 charge. This is because the transition metals have electrons in d subshell and do not follow the octet rule. Notice that there is no simple pattern for transition metal ions (or for the larger main group elements) as there is with the main group ions. \) shows the characteristic charges for some of these ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
